
Cobalt is a naturally occurring element in the earth's crust. It is a very small part of our environment. Cobalt is a component of vitamin B12, which supports the production of red blood cells. Very small amounts are needed for animals and humans to stay healthy. Cobalt poisoning can occur when you are exposed to large amounts of it. There are three basic ways that cobalt can cause poisoning. You can swallow too much of it, breathe too much into your lungs, or have it come in constant contact with your skin.Cobalt poisoning can also occur from the wear and tear of some cobalt/chromium metal-on-metal hip implants.
This type of implant is an artificial hip socket that is created by fitting a metal ball into a metal cup. Sometimes, metal particles (cobalt) are released as the metal ball grinds against the metal cup when you walk. These metal particles (ions) can get released into the hip socket and sometimes the bloodstream, causing cobalt toxicity.This article is for information only. DO NOT use it to treat or manage an actual poison exposure. If you or someone you are with has an exposure, call your local emergency number (such as 911), or your local poison center can be reached directly by calling the national toll-free Poison Help hotline (1-800-222-1222) from anywhere in the United States.
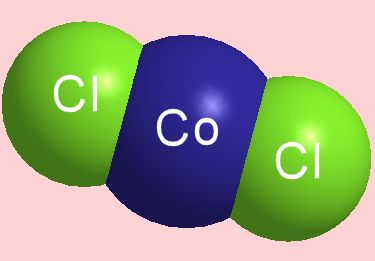
Cobalt chloride definition is - a chloride of cobalt; especially: the dichloride CoCl2 that is blue when dehydrated, turns red in the presence of moisture, and is used to indicate humidity. Cobalt(II) Chloride Hexahydrate CoCl2 6(H2O). CAS No.: 7791-13-1. Synonyms: Cobaltous chloride hexahydrate, Cobalt dichloride hexahydrate.
Usually you have to be exposed to high levels of cobalt for weeks to months to have symptoms. However, it is possible to have some symptoms if you swallow a large amount of cobalt at once.The most worrisome form of cobalt poisoning occurs when you breathe too much into your lungs. This usually will only happen in industrial settings where large amounts of drilling, polishing, or other processes release fine particles containing cobalt into the air.
Breathing in this cobalt dust can lead to chronic lung problems. If you breathe in this substance for long periods, you will likely develop breathing problems that are similar to asthma or pulmonary fibrosis, such as shortness of breath and decreased exercise tolerance.Cobalt poisoning that occurs from constant contact with your skin will likely cause irritation and rashes that go away slowly.Swallowing a large amount of absorbable cobalt at one time is very rare and is likely not very dangerous. It may cause nausea and vomiting. However, absorbing a large amount of cobalt over longer periods of time can lead to serious health problems, such as:. Cardiomyopathy (a problem where your heart becomes big and floppy and has problems pumping blood). Deafness. Nerve problems.
Ringing in the ears (tinnitus). Thickening of the blood. Thyroid problems. Vision problems. Your can be reached directly by calling the national toll-free Poison Help hotline (1-800-222-1222) from anywhere in the United States. This hotline will let you talk to experts in poisoning.
They will give you further instructions.This is a free and confidential service. All local poison control centers in the United States use this national number. You should call if you have any questions about poisoning or poison prevention. It does NOT need to be an emergency.
You can call for any reason, 24 hours a day, 7 days a week. If you swallowed a large amount of cobalt, or you are starting to feel sick from long-term exposure, you should go to an emergency room.Treatment for skin contact: Since these rashes are rarely serious, very little will be done. The area may be washed and a skin cream may be prescribed.Treatment for lung involvement: Breathing problems will be treated based on your symptoms. Breathing treatments and medications to treat swelling and inflammation in your lungs may be prescribed. Blood and urine tests, x-rays and ECG (electrocardiogram, or heart tracing) may be done.Treatment for swallowed cobalt: The health care team will treat your symptoms and order some blood tests. Blood and urine tests, x-rays and ECG (electrocardiogram, or heart tracing) may be performed. In the rare case that you have large levels of cobalt in your blood, you may need hemodialysis (kidney machine) and get medicines (antidotes) to reverse the effects of the poison.Treatment for signs of cobalt toxicity from a metal-on-metal hip implant may include removing the implant and replacing it with a traditional hip implant.
In: Aronson JK, ed. Meyler's Side Effects of Drugs. Waltham, MA: Elsevier; 2016:490-491.Lombardi AV, Bergeson AG. Evaluation of the failed total hip arthroplasty: history and physical examination. In: Scuderi GR, ed.
Techniques in Revision Hip and Knee Arthroplasty. Philadelphia, PA: Elsevier Saunders; 2015:chap 38.U.S. National Library of Medicine, Specialized Information Services, Toxicology Data Network website. Cobalt, elemental.
Updated September 5, 2017. Accessed January 17, 2019. For Health Content Provider (www.urac.org). URAC's is an independent audit to verify that A.D.A.M. Follows rigorous standards of quality and accountability. Is among the first to achieve this important distinction for online health information and services. Learn more about A.D.A.M.'
Is also a founding member of Hi-Ethics. This site complies with the HONcode standard for trustworthy health information:.The information provided herein should not be used during any medical emergency or for the diagnosis or treatment of any medical condition. A licensed physician should be consulted for diagnosis and treatment of any and all medical conditions. Call 911 for all medical emergencies. Links to other sites are provided for information only - they do not constitute endorsements of those other sites. Copyright 1997-2020, A.D.A.M., Inc. Duplication for commercial use must be authorized in writing by ADAM Health Solutions.
. Cobalt is a with the Co and atomic number 27. Like nickel, cobalt is found in the Earth's crust only in chemically combined form, save for small deposits found in alloys of natural. The, produced by reductive, is a hard, lustrous, silver-gray.Cobalt-based blue pigments have been used since ancient times for jewelry and paints, and to impart a distinctive blue tint to glass, but the color was later thought to be due to the known metal. Miners had long used the name ore (German for goblin ore) for some of the blue-pigment-producing; they were so named because they were poor in known metals, and gave poisonous -containing fumes when smelted.
In 1735, such ores were found to be reducible to a new metal (the first discovered since ancient times), and this was ultimately named for the kobold.Today, some cobalt is produced specifically from one of a number of metallic-lustered ores, such as (CoAsS). The element is, however, more usually produced as a by-product of and mining. In the (DRC) and yields most of the global cobalt production. World production in 2016 was 116,000 tonnes (according to ), and the DRC alone accounted for more than 50%.Cobalt is primarily used in, and in the manufacture of, wear-resistant and high-strength. The compounds cobalt silicate and (CoAl 2O 4, cobalt blue) give a distinctive deep blue color to,. Cobalt occurs naturally as only one stable, cobalt-59.
Is a commercially important radioisotope, used as a and for the production of high-energy.Cobalt is the active center of a group of called., the best-known example of the type, is an essential for all animals. Cobalt in inorganic form is also a for,. A block of refined cobalt (99.9% purity) cut from a large plateCobalt is a metal with a of 8.9. The is 1,115 °C (2,039 °F) and the magnetic moment is 1.6–1.7 per. Cobalt has a two-thirds that of.
Cobalt occurs as two:. The ideal transition temperature between the hcp and fcc structures is 450 °C (842 °F), but in practice the energy difference between them is so small that random intergrowth of the two is common.Cobalt is a weakly reducing metal that is protected from oxidation by a film. It is attacked. Heating in produces which loses oxygen at 900 °C (1,650 °F) to give the CoO.
The metal reacts with at 520 K to give; with and , producing equivalent binary. It does not react with or even when heated, but it does react with, and sulfur. At ordinary temperatures, it reacts slowly with, and very slowly with moist, but not with dry, air.Compounds. Cobalt(II) chloride hexahydrateFour of cobalt(II) are known: (CoF 2, pink), (CoCl 2, blue), (CoBr 2, green), (CoI 2, blue-black). These halides exist in anhydrous and hydrated forms. Whereas the anhydrous dichloride is blue, the hydrate is red.The reduction potential for the reaction Co 3 e − → Co 2+is +1.92 V, beyond that for to chloride, +1.36 V. Consequently, cobalt(III) and chloride would result in the cobalt(III) being reduced to cobalt(II).
Because the reduction potential for fluorine to fluoride is so high, +2.87 V, is one of the few simple stable cobalt(III) compounds. Cobalt(III) fluoride, which is used in some fluorination reactions, reacts vigorously with water. Coordination compounds As for all metals, molecular compounds and polyatomic ions of cobalt are classified as, that is, molecules or ions that contain cobalt linked to several. The principles of and of a series of ligands can be used to explain the usual oxidation state of cobalt. For example, Co 3+ complexes tend to have ligands. Because phosphorus is softer than nitrogen, phosphine ligands tend to feature the Co 2+ and Co +, an example being tris(triphenylphosphine)cobalt(I) chloride ((P(C 6H 5) 3) 3CoCl).
The more electronegative (and harder) oxide and fluoride can stabilize Co 4+ and Co 5+ derivatives, e.g. Caesium hexafluorocobaltate (Cs 2CoF 6) and potassium (K 3CoO 4)., a Nobel-prize winning pioneer in, worked with compounds of Co(NH 3) 6 3+. One of the isomers determined was. This coordination complex, a typical Werner-type complex, consists of a central cobalt atom coordinated by six orthogonal ligands and three counteranions. Using ligands in place of ammonia gives (Co(en) 3 3+), which was one of the first to be resolved into. The complex exists in the right- and left-handed forms of a 'three-bladed propeller'.
This complex was first isolated by Werner as yellow-gold needle-like crystals. Organometallic compounds. Main article:is a to, with cobalt in place of iron. Cobaltocene is much more sensitive to than ferrocene.
Cobalt carbonyl is a in and reactions. Vitamin B 12 (see ) is an organometallic compound found in nature and is the only that contains a metal atom. An example of an alkylcobalt complex in the otherwise uncommon +4 oxidation state of cobalt is the homoleptic complex (Co(1-norb) 4), a transition metal-alkyl complex that is notable for its stability to. The cobalt(III) and cobalt(V) complexes Li(THF) 4 +Co(1-norb) 4 − and Co(1-norb) 4 +BF 4 − are also known.

Isotopes. Main article:59Co is the only stable cobalt and the only that exists naturally on Earth. Twenty-two have been characterized: the most stable, has a of 5.2714 years; 57Co has a half-life of 271.8 days; 56Co has a half-life of 77.27 days; and 58Co has a half-life of 70.86 days.
All the other isotopes of cobalt have half-lives shorter than 18 hours, and in most cases shorter than 1 second. This element also has 4, all of which have half-lives shorter than 15 minutes.The isotopes of cobalt range in from 50 ( 50Co) to 73 u ( 73Co). The primary for isotopes with atomic mass unit values less than that of the most abundant stable isotope, 59Co, is and the primary mode of decay in isotopes with atomic mass greater than 59 atomic mass units is. The primary below 59Co are element 26 isotopes; above that the decay products are element 28 (nickel) isotopes. Early Chinese blue and white porcelain, manufactured c. 1335Cobalt compounds have been used for centuries to impart a rich blue color to,.
Cobalt has been detected in Egyptian sculpture, Persian jewelry from the third millennium BC, in the ruins of, destroyed in 79 AD, and in China, dating from the (618–907 AD) and the (1368–1644 AD).Cobalt has been used to color glass since the. The excavation of the yielded an ingot of blue glass, cast during the 14th century BC. Blue glass from Egypt was either colored with copper, iron, or cobalt. The oldest cobalt-colored glass is from the (1550–1292 BC). The source for the cobalt the Egyptians used is not known.The word cobalt is derived from the German kobalt, from meaning 'goblin', a superstitious term used for the of cobalt by miners. The first attempts to smelt those ores for copper or nickel failed, yielding simply powder (cobalt(II) oxide) instead.
Because the primary ores of cobalt always contain arsenic, smelting the ore oxidized the arsenic into the highly toxic and volatile, adding to the notoriety of the ore.Swedish chemist (1694–1768) is credited with discovering cobalt circa 1735, showing it to be a previously unknown element, distinct from bismuth and other traditional metals. Brandt called it a new 'semi-metal'. He showed that compounds of cobalt metal were the source of the blue color in glass, which previously had been attributed to the found with cobalt.
Cobalt became the first metal to be discovered since the pre-historical period. All other known metals (iron, copper, silver, gold, zinc, mercury, tin, lead and bismuth) had no recorded discoverers.During the 19th century, a significant part of the world's production of (a dye made with cobalt compounds and alumina) and ( powdered for use for pigment purposes in ceramics and painting) was carried out at the Norwegian.
The first mines for the production of smalt in the 16th century were located in Norway, Sweden, and Hungary. With the discovery of cobalt ore in in 1864, the mining of cobalt in Europe declined. With the discovery of ore deposits in, Canada in 1904 and the discovery of even larger deposits in the in the in 1914, the mining operations shifted again.
When the started in 1978, the copper mines of Katanga Province nearly stopped production. The impact on the world cobalt economy from this conflict was smaller than expected: cobalt is a rare metal, the pigment is highly toxic, and the industry had already established effective ways for recycling cobalt materials. In some cases, industry was able to change to cobalt-free alternatives.In 1938, John Livingood and discovered the radioisotope. This isotope was famously used at in the 1950s to establish violation in radioactive.After World War II, the US wanted to guarantee the supply of cobalt ore for military uses (as the Germans had been doing) and prospected for cobalt within the U.S.
An adequate supply of the ore was found in Idaho near in the side of a mountain. The firm Calera Mining Company started production at the site.It has been argued that cobalt will be one of the main objects of geopolitical competition in a world running on renewable energy and dependent on batteries, but this perspective has also been criticised for underestimating the power of economic incentives for expanded production. Occurrence The stable form of cobalt is produced in through the. It comprises.
Free cobalt (the ) is not found on Earth because of the oxygen in the atmosphere and the chlorine in the ocean. Both are abundant enough in the upper layers of the Earth's crust to prevent native metal cobalt from forming. Except as recently delivered in meteoric iron, pure cobalt in native metal form is unknown on Earth. The element has a medium abundance but natural compounds of cobalt are numerous and small amounts of cobalt compounds are found in most rocks, soils, plants, and animals.In nature, cobalt is frequently associated with.
Both are characteristic components of, though cobalt is much less abundant in iron meteorites than nickel. As with nickel, cobalt in meteoric iron may have been well enough protected from oxygen and moisture to remain as the free (but alloyed) metal, though neither element is seen in that form in the ancient terrestrial crust.Cobalt in compound form occurs in copper and nickel minerals.
It is the major metallic component that combines with and arsenic in the sulfidic (CoAsS), (CoAs 2), ((Co,Fe)AsS), and (CoAs 3) minerals. The mineral is similar to and occurs together with in the copper deposits of. When it reaches the atmosphere, occurs; the sulfide minerals oxidize and form pink ('cobalt glance': ) and (CoCO 3).Cobalt is also a constituent of. The readily absorbs and accumulates like cobalt from the surrounding soil in its leaves. These are subsequently inhaled during.
Production. See also: Cobalt mine production (2017) and reserves in tonnes according to CountryProductionReserves64,0003,500,0005,6,0001,200,0004,3,2,0,8,9,800-2,5023,000Other countries5,900560,000World total110,0007,100,000The main ores of cobalt are, erythrite, and (see above), but most cobalt is obtained by reducing the cobalt of nickel and mining and.Since cobalt is generally produced as a by-product, the supply of cobalt depends to a great extent on the economic feasibility of copper and nickel mining in a given market.
Demand for cobalt was projected to grow 6% in 2017.Several methods exist to separate cobalt from copper and nickel, depending on the concentration of cobalt and the exact composition of the used. One method is, in which bind to ore components, leading to an enrichment of cobalt ores. Subsequent converts the ores to, and the copper and the iron are oxidized to the oxide.
With water extracts the sulfate together with the. The residues are further leached with, yielding a solution of copper sulfate. Cobalt can also be leached from the of copper smelting.The products of the above-mentioned processes are transformed into the cobalt oxide (Co 3O 4). This oxide is reduced to metal by the or reduction with carbon in a.
Extraction The estimates world reserves of cobalt at 7,100,000 metric tons. The (DRC) currently produces 63% of the world's cobalt. This market share may reach 73% by 2025 if planned expansions by mining producers like Plc take place as expected. But by 2030, global demand could be 47 times more than it was in 2017, Bloomberg New Energy Finance has estimated.Changes that Congo made to mining laws in 2002 attracted new investments in Congolese copper and cobalt projects. Glencore's shipped 24,500 tons of cobalt in 2016, 40% of Congo DRC's output and nearly a quarter of global production. After oversupply, Glencore closed Mutanda for two years in late 2019. Glencore's project is resuming as well and should produce 300,000 tons of copper and 20,000 tons of cobalt by 2019, according to Glencore.
Democratic Republic of the Congo. See also:, andIn 2005, the top producer of cobalt was the copper deposits in the 's. Formerly Shaba province, the area had almost 40% of global reserves, reported the in 2009. By 2015, Democratic Republic of the Congo (DRC) supplied 60% of world cobalt production, 32,000 tons at $20,000 to $26,000 per ton. Recent growth in production could at least partly be due to how low mining production fell during DRC Congo's very violent civil wars in the early 2000s, or to the changes the country made to its Mining Code in 2002 to encourage foreign and multinational investment and which did bring in a number of investors, including.supplied 10% to 25% of the DRC production. Some 100,000 cobalt miners in Congo DRC use hand tools to dig hundreds of feet, with little planning and fewer safety measures, say workers and government and NGO officials, as well as reporters' observations on visits to isolated mines.
The lack of safety precautions frequently causes injuries or death. Mining pollutes the vicinity and exposes local wildlife and indigenous communities to toxic metals thought to cause birth defects and breathing difficulties, according to health officials.Human rights activists have alleged, and reported confirmation, that is used in mining cobalt from African.
This revelation prompted cell phone maker, on March 3, 2017, to stop buying ore from suppliers such as who source from artisanal mines in the DRC, and begin using only suppliers that are verified to meet its workplace standards.The political and ethnic dynamics of the region have in the past caused outbreaks of violence and years of armed conflict and displaced populations. This instability affected the price of cobalt and also created perverse incentives for the combatants in the First and Second Congo Wars to prolong the fighting, since access to diamond mines and other valuable resources helped to finance their military goals—which frequently amounted to genocide—and also enriched the fighters themselves. While DR Congo has in the 2010s not recently been invaded by neighboring military forces, some of the richest mineral deposits adjoin areas where Tutsis and Hutus still frequently clash, unrest continues although on a smaller scale and refugees still flee outbreaks of violence.Cobalt extracted from small Congolese endeavors in 2007 supplied a single Chinese company, Congo DongFang International Mining. A subsidiary of, one of the world's largest cobalt producers, Congo DongFang supplied cobalt to some of the world's largest battery manufacturers, who produced batteries for ubiquitous products like the Apple. Corporate pieties about an ethical were thus met with some incredulity.
A number of observers have called for tech corporations and other manufacturers to avoid sourcing conflict metals in Central Africa at all rather than risk enabling the financial exploitation, abuses like kidnappings for, environmental devastation and the human toll of violence, poverty and toxic conditions.The project, operated by the (CAMEC) in, may be the richest cobalt reserve in the world. It produced an estimated one-third of the total global cobalt production in 2008. In July 2009, CAMEC announced a long-term agreement to deliver its entire annual of cobalt concentrate from Mukondo Mountain to Zhejiang Galico Cobalt & Nickel Materials of China.In February 2018, global asset management firm defined the DRC as economically 'the of the electric vehicle age,' due to its cobalt resources, as essential to the that drive.On March 9, 2018, President updated the 2002 mining code, increasing royalty charges and declaring cobalt and 'strategic metals'. Canada In 2017, some exploration companies were planning to survey old silver and cobalt mines in the area of where significant deposits are believed to lie.
The mayor of Cobalt stated that the people of Cobalt welcomed new mining endeavours and pointed out that the local work force is peaceful and English-speaking, and good infrastructure would allow much easier sourcing of spare parts for the equipment or other supplies than were to be found in conflict zones.Applications In 2016, 116,000 tonnes of cobalt was used.Cobalt has been used in the production of high-performance alloys. It can also be used to make rechargeable batteries, and the advent of electric vehicles and their success with consumers probably has a great deal to do with the DRC's soaring production. Other important factors were the 2002 Mining Code, which encouraged investment by foreign and transnational corporations such as Glencore, and the end of the First and Second Congo Wars.Alloys Cobalt-based have historically consumed most of the cobalt produced. The temperature stability of these alloys makes them suitable for turbine blades for and aircraft, although nickel-based alloys surpass them in performance. Cobalt-based alloys are also - and wear-resistant, making them, like, useful for making orthopedic that don't wear down over time. The development of wear-resistant cobalt alloys started in the first decade of the 20th century with the alloys, containing chromium with varying quantities of tungsten and carbon.
Alloys with and are very hard and wear-resistant. Special cobalt-chromium- alloys like are used for parts (hip and knee replacements).
Cobalt alloys are also used for prosthetics as a useful substitute for nickel, which may be allergenic. Some also contain cobalt for increased heat and wear resistance.
The special alloys of aluminium, nickel, cobalt and iron, known as, and of samarium and cobalt are used in. It is also alloyed with 95% for jewelry, yielding an alloy suitable for fine casting, which is also slightly magnetic. Batteries (LiCoO 2) is widely used in cathodes. The material is composed of cobalt oxide layers with the lithium. During dischargethe lithium is released as lithium ions. (NiCd) and (NiMH) batteries also include cobalt to improve the oxidation of nickel in the battery.Transparency Market Research estimated the global lithium-ion battery market at $30 billion in 2015 and predicted an increase to over US$75 billion by 2024.Although in 2018 most cobalt in batteries was used in a mobile device, a more recent application for cobalt is rechargeable batteries for electric cars. This industry has increased five-fold in its demand for cobalt, which makes it urgent to find new raw materials in more stable areas of the world.
Demand is expected to continue or increase as the prevalence of electric vehicles increases. Exploration in 2016–2017 included the area around, an area where many silver mines ceased operation decades ago. Cobalt for electric vehicles increased 81% from the first half of 2018 to 7,200 tonnes in the first half of 2019, for a battery capacity of 46.3 GWh. The future of electric cars may depend on deep-sea mining, since cobalt is abundant in rocks on the seabed.Since child and slave labor have been repeatedly reported in cobalt mining, primarily in the artisanal mines of DR Congo, technology companies seeking an ethical supply chain have faced shortages of this raw material and the price of cobalt metal reached a nine-year high in October 2017, more than US$30 a pound, versus US$10 in late 2015. After oversupply, the price dropped to a more normal $15 in 2019. Catalysts Several cobalt compounds are oxidation catalysts. Cobalt acetate is used to convert to, the precursor of the bulk polymer.
Typical catalysts are the cobalt (known as cobalt soaps). They are also used in paints, varnishes, and inks as 'drying agents' through the oxidation of. The same carboxylates are used to improve the adhesion between steel and rubber in steel-belted radial tires. In addition they are used as accelerators in systems.Cobalt-based catalysts are used in reactions involving.
Cobalt is also a catalyst in the for the of carbon monoxide into liquid fuels. Of often uses as a catalyst, although it is often replaced by more efficient iridium- and rhodium-based catalysts, e.g. The.The of uses a catalyst derived from cobalt and molybdenum. This process helps to clean petroleum of sulfur impurities that interfere with the refining of liquid fuels. Pigments and coloring. Cobalt-colored glassBefore the 19th century, cobalt was predominantly used as a pigment. It has been used since the Middle Ages to make, a blue-colored glass.
Smalt is produced by melting a mixture of roasted mineral, and, which yields a dark blue silicate glass, which is finely ground after the production. Smalt was widely used to color glass and as pigment for paintings. In 1780, discovered, and in 1802 discovered cobalt blue.
Cobalt pigments such as (cobalt aluminate), blue (cobalt(II) stannate), various hues of (a mixture of and ), and cobalt violet are used as artist's pigments because of their superior chromatic stability. (cobalt yellow) is now largely replaced by more lightfast yellow pigments.Radioisotopes (Co-60 or 60Co) is useful as a gamma-ray source because they can be produced in predictable quantity and high by bombarding cobalt with. It produces with energies of 1.17 and 1.33.Cobalt is used in, sterilization of medical supplies and medical waste, radiation treatment of (cold ), (e.g. Weld integrity radiographs), density measurements (e.g. Concrete density measurements), and tank fill height switches.
The metal has the unfortunate property of producing a fine dust, causing problems with. Cobalt from radiotherapy machines has been a serious hazard when not discarded properly, and one of the worst radiation contamination accidents in North America occurred in 1984, when a discarded radiotherapy unit containing cobalt-60 was mistakenly disassembled in a junkyard in Juarez, Mexico.Cobalt-60 has a radioactive half-life of 5.27 years. Loss of potency requires periodic replacement of the source in radiotherapy and is one reason why cobalt machines have been largely replaced by in modern radiation therapy. (Co-57 or 57Co) is a cobalt radioisotope most often used in medical tests, as a radiolabel for vitamin B 12 uptake, and for the. Cobalt-57 is used as a source in and is one of several possible sources in devices.could intentionally incorporate 59Co, some of which would be activated in a to produce 60Co. The 60Co, dispersed as, is sometimes called a.
Other uses. Cobalt is used in for its attractive appearance, hardness, and resistance to. It is also used as a base primer coat for.Biological role.
Cobalt-deficient sheepCobalt is essential to the metabolism of all. It is a key constituent of, also known as vitamin B 12, the primary biological reservoir of cobalt as an. In the stomachs of animals convert cobalt salts into vitamin B 12, a compound which can only be produced by bacteria. A minimal presence of cobalt in soils therefore markedly improves the health of animals, and an uptake of 0.20 mg/kg a day is recommended because they have no other source of vitamin B 12.Proteins based on cobalamin use to hold the cobalt. Coenzyme B 12 features a reactive C-Co bond that participates in the reactions.
In humans, B 12 has two types of: and adenosyl. Promotes methyl (−CH 3) group transfers.
The adenosyl version of B 12 catalyzes rearrangements in which a hydrogen atom is directly transferred between two adjacent atoms with concomitant exchange of the second substituent, X, which may be a carbon atom with substituents, an oxygen atom of an alcohol, or an amine. (MUT) converts to, an important step in the extraction of energy from proteins and fats.Although far less common than other (e.g. Those of zinc and iron), other cobaltoproteins are known besides B 12. These proteins include, an enzyme that occurs in humans and other mammals that does not use the corrin ring of B 12, but binds cobalt directly. Another non-corrin cobalt enzyme is, an enzyme in bacteria that metabolizes. Cobalt deficiency in animals In the early 20th century, during the development of farming on the of New Zealand, cattle suffered from what was termed 'bush sickness'. It was discovered that the volcanic soils lacked the cobalt salts essential for the cattle food chain.The 'coast disease' of sheep in the of the of in the 1930s was found to originate in nutritional deficiencies of trace elements cobalt and copper.
The cobalt deficiency was overcome by the development of 'cobalt bullets', dense pellets of cobalt oxide mixed with clay given orally for lodging in the animal's. Health issues.
Cobalt is an essential element for life in minute amounts. The value for soluble cobalt salts has been estimated to be between 150 and 500 mg/kg. In the US, the (OSHA) has designated a (PEL) in the workplace as a time-weighted average (TWA) of 0.1 mg/m 3. The (NIOSH) has set a (REL) of 0.05 mg/m 3, time-weighted average. The (immediately dangerous to life and health) value is 20 mg/m 3.However, chronic cobalt ingestion has caused serious health problems at doses far less than the lethal dose. In 1966, the addition of cobalt compounds to stabilize in Canada led to a peculiar form of toxin-induced, which came to be known as beer drinker's cardiomyopathy.Furthermore, cobalt metal is suspected of causing (i.e., possibly, ) as per the (IARC) Monographs.It causes respiratory problems when inhaled.
It also causes skin problems when touched; after and chromium, cobalt is a major cause of. These risks are faced by cobalt miners.Cobalt can be effectively absorbed by charred pigs' bones; however, this process is inhibited by copper and zinc, which have greater affinities to bone char. See also.References.

